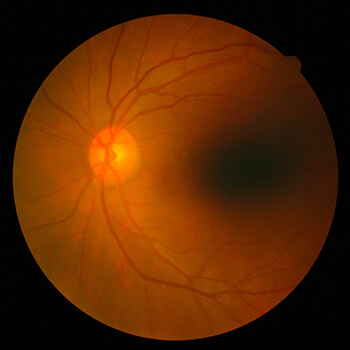
Importance of Retina Eye Exams
The retina is an extension of the brain. Much like the brain tissue, the retina cannot regenerate. Diseases of the retina can cause permanent blindness; therefore, having regular eye exams is critical to preserve your vision. Our Inland Empire retina specialists perform retina exams in order to diagnose problems as early as possible so treatment can be administered to stop disease progression. Retinal eye exams are especially important for diabetic patients.
Common Retina Conditions
- Diabetic Eye Care
- Retinal Detachment
- Age-Related Macular Degeneration
- Macular Hole/Pucker
- Retinal Vein and Artery Occlusion
Diabetic Eye Care
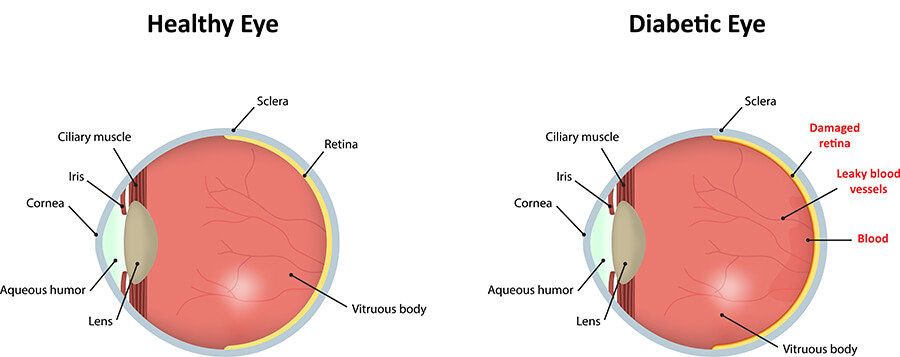
Diabetic retinopathy can occur in both Type 1 and Type 2 diabetics. The longer a person has had diabetes, the higher their chances of having some form of diabetic retinopathy. Retinopathy is present in 90% of those who have had the disease for more than 20 years.
Diabetic retinopathy damages the blood vessels in the retina. As the blood vessels become weak, they can rupture, leading to a reduction in blood flow to the retina. To compensate, the retina will try to grow new blood vessels to replace the damaged ones.
Unfortunately, these new blood vessels usually grow in the wrong places. They are fragile and they break easily, sometimes spilling enough blood to fill up the eye. When these new blood vessels begin to grow, it is called proliferative diabetic eye disease.
Symptoms of Diabetic Retinopathy
Because this is a progressive disease, there are usually no symptoms in the early stages. By the time you experience vision problems, the disease will be in a later stage of development. If you are diabetic and are experiencing any of the following symptoms, contact our diabetic eye doctors immediately:
- Dark spots
- Floaters or strings in vision
- Vision loss
- Blurry vision
- Changes in color perception
Treatment for Diabetic Retinopathy
Laser treatment can prevent or delay severe visual loss from diabetic retinopathy, but only if it’s diagnosed early enough. Regular eye exams are critical for detecting this disease early so treatment can be administered and vision preserved.
A diabetic can significantly reduce the risk of developing diabetic retinopathy by adopting a healthy lifestyle and making eye health a priority:
- See an eye doctor for a dilated diabetic eye exam at least once a year
- Work with your primary care physician to monitor blood pressure and blood sugar levels and keep them under control
- Maintain a healthy diet
- Exercise regularly
Retinal Detachment
A retinal detachment is a serious eye condition in which the retina detaches from the back of the eye. This signifies a medical emergency, so if you experience any of the symptoms below, contact our Inland Empire retinal specialists immediately.
Symptoms of Retinal Detachment
- Specks in your vision that look like cobwebs
- Flashes in your eyesight
- Wavy vision
- A dark shadow over your vision
- Loss of vision
Common Causes of Retinal Detachment
- The contraction of vitreous gel inside the eye
- A retinal tear that causes fluid to get underneath the retina
- Trauma or injury to the eye that causes fluid to collect under the retina
- The contraction of scar tissue on the retina
Treatments for Retinal Detachment
Our retinal doctors will assess the root cause of your retina problem in order to recommend the most effective treatment. Treatment may include different procedures to reduce retinal pulling due to vitreous gel contractions such as a vitrectomy to remove part of the vitreous gel from the eye. Laser surgery may help to reconnect the retina. Swift and accurate treatment will be vital to preserving your vision.
Age-Related Macular Degeneration
Age-related macular degeneration (AMD) is a progressive retina disease that affects the center of the retina, the macula. The macula is responsible for central vision, so any damage to this area will affect your ability to perform daily tasks.
Types of Age-Related Macular Degeneration
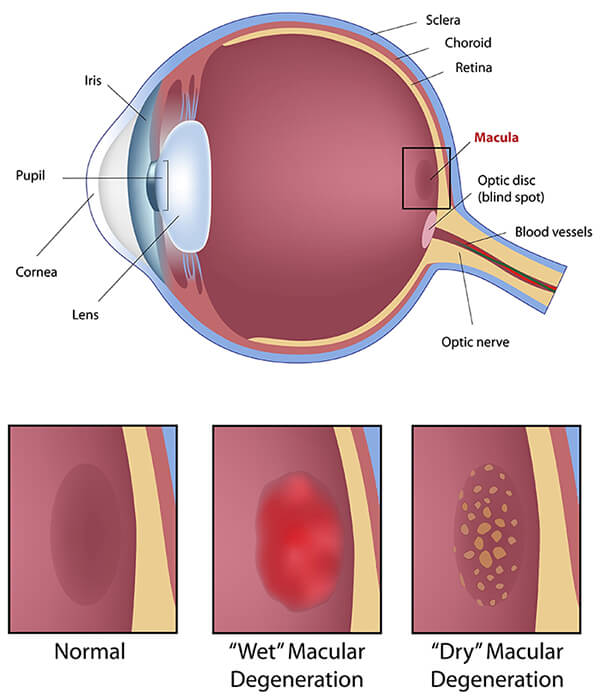
Symptoms of Macular Degeneration
- Distortion (straight lines or objects appear wavy or crooked)
- Blurriness
- Dark areas
- Permanent loss of central vision
- Peripheral vision remains clear
Age-Related Macular Degeneration Treatments
We offer multiple treatment options for Macular Degeneration based upon your needs and current visual situation. The best way to determine what treatment options would be best for you is to schedule a dilated eye exam with our Macular Degeneration Specialists.
Age Related Macular Degeneration Treatment Options
- Laser Treatment
- Antiangiogenic Drugs Like Lucentis and Avastin
- EEYLEA® Treatment
- Vitamins (AREDS)
Antiangiogenic Drugs
Antiangiogenic drugs, like Lucentis and Avastin, work to help shrink existing abnormal or weak blood vessels and prevent the growth of new leaky blood vessels that cause wet macular degeneration.
Macular Hole and Macular Pucker
The macula is a small area in the center of the retina where light is sharply focused to produce the detailed color vision needed for tasks such as reading and driving. When a full-thickness defect develops in the macula, the condition is referred to as a macular hole. Two conditions called a macular pucker and a macular hole can cause vision to become blurred or distorted, affecting central vision.
Symptoms of Macular Hole or Macular Pucker
- Blurring
- Distortion (straight lines appearing wavy)
- A dark spot in the central vision
The degree to which vision is affected will depend on the size and location of the macular hole as well as the stage of its development.
Treatment for Macular Holes and Macular Puckers
In some situations, the macular hole will correct itself and treatment will not be necessary. In more severe cases, we might recommend a vitrectomy procedure to remove the vitreous gel from the eye and replace it with a gas bubble to minimize tugging on the retina tissue.















